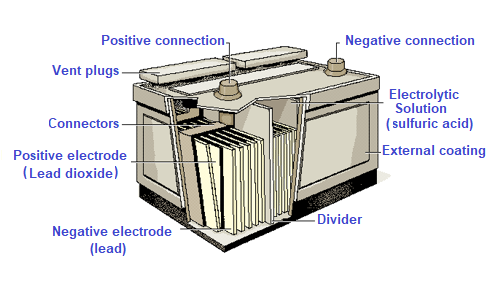
Battery capacity A battery could be any device that stores energy for later use. The word battery identifies an electrochemical apparatus that converts chemical energy into electrical energy through the use of a galvanic cell.
A galvanic cell consists of two electrodes (an anode and a cathode) and an electrolyte solution. The voltage of a galvanic cell of a lead battery is typically 2 Volts and consists of one or more galvanic cells. The purpose of batteries is not to produce electricity but to accumulate or store electricity. The chemical reactions inside the batteries vary depending on whether the electrical energy is stored or released. In rechargeable batteries such as lead-acid batteries, the process can be repeated over and over again. Typically, batteries do not offer 100% efficiency but are at lower values. This means that some energy is lost in the form of heat during the chemical reactions of charge and discharge.
Nominal Voltage The nominal voltage (Volt) is the potential difference established between the positive and negative poles. As an example, I report the rated voltage, at a temperature of 25 ° C, of the most common accumulators:
The rated voltage is equal to the sum of the rated voltages that make up the accumulator. During use, the battery voltage is not constant but undergoes variations due to the charging and discharging process, and to protect the battery pack; some Voltage limits are set where must be contained variations. In the charging process, the maximum value of voltage sent to the battery pack is equal to the charging voltage, while the minimum value during discharge is given by the discharge voltage.
Charging voltage The charge voltage is expressed in Volts and supplied by the battery, which allows triggering the recombination process of the electric charges. The manufacturer indicates different charging modes. For example, lead and lithium batteries need a constant charging voltage, while nickel batteries require a constant current, so they must not exceed the specific values; otherwise, hydrogen gas is produced in lead-acid batteries and probable damage, and there is a risk of explosion in lithium batteries. As an example, I report the limit values of the charging voltage, at a temperature of 25 ° C, of the most common accumulators:
The charging voltage varies as the accumulator temperature changes, so it needs compensation. If the temperature is different from 25 ° C, it needs compensation. For example, for lead batteries, the charge limit voltage at a temperature of 15 ° C becomes 2.445 Volt per element and 2.335 at a temperature of 35 ° C. Final landfill voltage The final discharge voltage is that indicated by the manufacturer and is expressed in Volts. To avoid reducing the accumulator life, it is necessary not to discharge it beyond the voltage limit indicated by the manufacturer. Example. An accumulator with a voltage of 12 Volts and a Capacity of 10 Ah, the discharge current at 0.05 C and 2 C will be expressed by the following rule: 0.05C = 0.05 * 10 = 0.5A and 2C = 2 * 10 = 20A Where the C index is the intensity of the current that a battery delivers to discharge in an hour Note: The C index indicates the current intensity in [Ah] delivered by the battery in the case of discharge in 1 hour, C5 in the case of discharge in 5 hours, C20 in 20 hours, etc. A 2C index represents the intensity of current in the case of discharge in ½ hour. An accumulator with a nominal capacity Q equal to 200 Ah and an index C₁₀, can deliver for 10 hours (at a constant temperature of 25 ° C) a nominal current of 20 A. In the same way, if the same accumulator has an index C₂₀ it can deliver for 20 hours a discharge current of 10 A. Capacity The battery's capacity is the parameter that represents the amount of electrical charge which is stored in a battery and is expressed in Ampere-hours (Ah). The Ampere-hour (Ah) is the unit of measurement of the electric charge and represents the amount of charge required to deliver 1 Ampere for one hour, so it is used to measure the discharge time of a battery (e.g. a 1 Ah battery if it continuously delivers 1 A in 1 hour), but it is also used to estimate the charging time (e.g. to charge a battery from 1 Ah with a constant current of 1 A it would take 1 hour) Calculation of capacity The nominal capacity (Q) of an accumulator (battery) at an ambient temperature of 25 ° C is equal to the product of the discharge current (A) and the time (h) that is required to reach the final discharge voltage. Basically, it indicates the amount of electrical charge that an electrochemical cell can deliver before discharging. Q = Is * t Q = nominal capacity (unit of measurement: Ah); Is = discharge current (A); t = time (h) The nominal capacity Q (defined to homogenize the criteria for choosing an accumulator) is conventionally indicated at the discharge in a determined time interval (h) and refers to a constant discharge current. The value of the capacity of a battery does not remain constant but is influenced by two main factors: the temperature and the intensity of the discharge current. If the temperature will rises, as a consequence, the capacity increases and vice versa; if the discharge current will increases, the capacity decreases and vice versa. The energy of a battery (Wh = watt-hour) The unit of measurement of the energy storage capacity of a battery is the watt-hour (Wh). It is obtained by multiplying the capacity in Ah (ampere-hour) by the nominal voltage of the battery: Wh = V * Ah Example: 12 Volt accumulator voltage; Capacity 50 Ah. Wh = 50 (Ah) * 12 (Volt) = 600
Ampere-hour calculation If I wanted to calculate the Ah of a battery that delivers 3 kWh at 12 Volts, I would do this to obtain the Energy expressed in Wh I have to multiply the value of the capacity in Ah by the rated voltage Vn of the storage system: Wh = Ah * Vn From which I get the Ah with the formula: Ah = Wh / Vn So: Ah = 3000/12 = 250 Example: To get 100 Watts for 10 hours from a 12 Volt (V) battery, you will need: 83.3 Ah (100 * 10/12 = 83.3 Ah). From the value obtained, it must take into account that if you use lead-acid batteries or lead gel, they must be discharged to 50% of the nominal capacity or 80% if they are lithium, so it is necessary to increase the capacity. In the case of lead-acid batteries, I divide the Ah by 0.5; and for 0.8 in the case of lithium batteries. So if I have to use a lead-acid battery with a nominal capacity (Q) of 200 Ah, I will have to buy one of 400 Ah (Ah = Q / 0.5 = 200 / 0.5 = 400 Ah), if instead, with battery Lithium with a nominal capacity of 200 Ah I will have to buy a 250 Ah one (Ah = Q / 0,8 = 200 / 0,8 = 250 Ah)
Standard values of a completely discharged battery Standard means: the nominal capacity of a battery (Q) with an index C₂₀, is the amount of current that the battery can supply continuously for 20 hours before dropping to 10.5 Volts which is the standard value of a completely discharged battery.
The standard value of a fully charged battery Instead, it is fully charged, when the voltage at the battery poles is about 12.8 Volts.
A lead-acid battery with elements immersed in sulfuric acid can withstand discharges not exceeding 50% of its nominal capacity. If it is exceeded, it can be irreversibly damaged. For Gel batteries, the value is slightly higher. So if the battery has a capacity of 100 Ah, it will only be able to supply 50 Ah before it is discharged.
|
About
We offer the best and economical solutions, backed by 27+ years of experience and international standards knowledge, echnological changes, and industrial systems.