Aluminium Special Alloy
Aluminium is produced from the ore, Bauxite. The bauxite is made into alumina powder and is then smelted at a high temperature to produce aluminium metal. Aluminium is fully recyclable without losing its properties or qualities, even with repeated recycling sequences. Aluminium is the most plentiful metal on earth; it is lightweight it has a low density, is non-toxic, has high thermal and electrical conductivity and does not spark. Aluminium contains no iron (Fe) so is not magnetisable, but it is paramagnetic. Aluminium will be weakly attracted to an externally applied magnetic field, however, this will not be maintained once this field is removed; no magnetism will remain.
Aluminium is often the best solution when reduced weight, good conductivity, and high formability are desirable characteristics. For this reason, knowing all the aluminium processing steps perfectly is truly important.
Read further and learn more about:
-
What is Aluminum?
-
Aluminum Grades
-
Applications of Aluminum Grades
-
Types of Aluminum Products
For more details consult the Aluminium Series Information Guides in the figures below, which include the datasheet for each type.
What is Aluminum?
Aluminum is the most abundant metal in Earth’s crust but rarely exists in elemental form. The various forms of aluminium and its alloys are valued for their low density and high strength-to-weight ratio, durability, and corrosion resistance. Since aluminium is 2.5 times less dense than steel, it is an excellent alternative to steel in applications requiring mobility and portability.
Many aluminium alloys are ductile and malleable, making them easy to form and machine. They are good electrical and thermal conductors with non-sparking and non-magnetic properties. Aluminium is recyclable, with a low re-melting temperature that requires 5% of the energy needed to produce the primary metal. Seventy-five percent of aluminium can be recovered for reuse without losing its properties, which makes aluminium sustainable and environmentally friendly.
Aluminium Grades
Pure aluminium is combined with different alloying elements to modify its mechanical properties, corrosion resistance, and formability and machinability, which determines the various grades. The Aluminum Association created and is responsible for maintaining the nomenclature for the standard aluminium grades, which are categorized according to their main alloying element and mechanical and thermal treatment response.
There are two main classifications of aluminium alloys: wrought and cast aluminium. Each classification has a different identification numbering system to distinguish it. Wrought and cast aluminium are differentiated by how they are processed, with cast aluminium being melted and poured into a mould, while wrought aluminium is worked in solid form.
The different manufacturing processes produce grades of aluminium alloys with unique properties. The classifications add to the difficulty of determining which grade of aluminium to use for a project. Cast aluminium has a higher percentage of alloying material, while wrought aluminium has greater tensile strength.
Applications of Aluminum Grades
Pure aluminium is combined with different alloying elements to modify its mechanical properties, corrosion resistance, and formability and machinability, which determines the various grades. The Aluminum Association created and is responsible for maintaining the nomenclature for the standard aluminium grades, which are categorized according to their main alloying element and mechanical and thermal treatment response.
There are two main classifications of aluminium alloys: wrought and cast aluminium. Each classification has a different identification numbering system to distinguish it. Wrought and cast aluminium are differentiated by how they are processed, with cast aluminium being melted and poured into a mould, while wrought aluminium is worked in solid form.
The different manufacturing processes produce grades of aluminium alloys with unique properties. The classifications add to the difficulty of determining which grade of aluminium to use for a project. Cast aluminium has a higher percentage of alloying material, while wrought aluminium has greater tensile strength.
Types of Aluminum Products
Wrought aluminium has exceptional mechanical strength and can be formed into many shapes. It is produced by smelting aluminium ingots with a measured amount of an alloying metal, resulting in the grade's composition. The smelted aluminium alloy is cast into billets or slabs and mechanically processed by rolling, forging, or extrusion. Heat treatment further improves the aluminium alloy's natural properties.
The advantages of wrought aluminium include:
- Exceptional mechanical properties
- Structural integrity without defects
- Smooth, even surface finish
- Excellent weldability and machinability
- Formability – can be transformed, shaped, machined, and extruded into any possible shape.
A four-digit number code identifies each wrought aluminum grade:
- The first digit indicates the primary alloying element added to the pure aluminum. The primary alloying element affects the grade’s properties in a series.
- The second digit refers to the modification of an alloy. The modifications are registered with the IADS, which requires specific documentation. If the designated number is zero, the alloy is original or unmodified.
- The third and fourth digits are arbitrary numbers assigned to a specific alloy in the series. For the 1000 series, these digits indicate the purity of the alloy.
The table below summarizes the wrought aluminium series. Series 1000 is the purest form of aluminium with the lowest yield and tensile strength, while the 7000 series, with alloys of magnesium, zinc, and copper, has the highest tensile and yield strength.
| Temper | Composition | Tensile Strength (MPa) | Yield Strength (MPa) |
| 1000 Series | 99.00%-99.99% Aluminum | 82-166 | 28-152 |
| 2000 Series | 2.2%-6.8% Copper | 110-283 | 41-248 |
| 3000 Series | 0.3%-1.5% Manganese | 110-283 | 41-248 |
| 4000 Series | 3.6%-13.5% Silicon | 172-414 | 45-180 |
| 0.1%-4.7% Copper | |||
| 0.05%-5.5% Magnesium | |||
| 5000 Series | 0.05%-5.5% Magnesium | 124-352 | 41-345 |
| 6000 Series | 0.2%-18% Silicon | 124-310.3 | 55.2-276 |
| 0.35%-1.5% Magnesium | |||
| 7000 Series | 0.8%-8.2% Zinc | 228-572 | 103-503 |
| 0.1%-3.4% Magnesium | |||
| 0.05%-2.6% Copper |
Aluminium Corrosion resistance
The corrosion behaviour of Aluminium & its alloys is controlled by the fact that like stainless steel it is a passive metal with a protective air-formed film forming naturally on the surface. This naturally forming film is made up of two layers which normally have a total thickness of about 7 nanometers. The lowest layer is termed the barrier layer & it is amorphous and has no structure. The layer that grows on top of this is hydrated & less compact. It tends to grow at high humidities or with contact with water.
The air-formed passive layers are very stable & result in very low corrosion rates in neutral environments. However, the film dissolves in both acid & alkaline conditions. High corrosion rates can be expected below pH 3 & above pH 9. Figure below shows what happens when aluminium corrodes

With knowledge of all the aluminium alloys available on the market, it is possible to avoid these unpleasant situations. The list below shows all Aluminium series
- 1000 Series Aluminium alloys are variations on pure aluminium. They are unalloyed & are generally more than 99% pure & have a combination of good electrical conductivity & chemical resistance.
- 2000 Series Aluminium al6000 Series Aluminium alloys contain both magnesium & silicon. they have good resistance to atmospheric corrosion and form an attractive film after anodising. They are relatively easy to roll extrude and forge.loys contain copper as the main alloying ingredient. They have moderately high strength but tend to be brittle & exhibit poorer corrosion resistance than other alloy series.
- 3000 Series Aluminium alloys containing silicon in combination with copper or magnesium. They con be age-hardened to improve their mechanical properties. They have fairly good corrosion resistance but are particularly resistant to pitting corrosion.
- 4000 Series Aluminium alloys also contain silicon as the main alloying ingredient but do not contain copper or magnesium. Their main use stems from their lower melting point which confers good casting qualities.
- 5000 Series Aluminium alloys are based around magnesium. They have good resistance to sea water & find use in marine applications as they have both moderate strength & ductility. they are easy to weld & anodize well.
- 6000 Series Aluminium alloys contain both magnesium & silicon. they have good resistance to atmospheric corrosion and form an attractive film after anodising. They are relatively easy to roll extrude and forge.
- 7000 Series Aluminium alloys contain zinc as the majority alloying metal but may also contain additions of copper, magnesium chromium or zirconium. Some of the copper-containing variants have a strong tenancy to exfoliation corrosion in the heat-affected zone of welds. However, allots such as 7075 have good resistance to aggressive atmospheres.
- The 8000 series Aluminium alloys may contain tin, iron or silicon. They are relatively uncommon but can be rolled to thin sheets which gives them some use as fins for heat exchangers & as dishes or thin foils.
As you can see from the list above, each series has different features, so choosing the right one is of fundamental importance.
There are several type of corrosion, all described below:
Uniform Corrosion & Passive Films
Several metals such as iron, chromium, and aluminium are inherently reactive because they occur in nature as their ores rather than in elemental form. However, these metals and their alloys are widely used in industry because, under particular environmental conditions, they lose their chemical reactivity and become inert. This phenomenon, which is known as passivity, is a result of the reaction of the metal with water and/or oxygen to form a highly adherent and very thin oxide film on the metal surface. A familiar example of passive alloys is stainless steel, which is used in different applications from kitchenware to the petrochemical industry. Passivity affects the kinetics of the corrosion process (i.e., corrosion rate) by producing a protective film that acts as a barrier against corrosion attacks. However, passive films are not perfect and certain conditions lead to the breakdown of passivity, and hence, localized corrosion. Pitting, crevice and stress corrosion cracking are the main forms of localized corrosion, which directly or indirectly cause corrosion failures of industrial assets. Figure 2 below shows all dettails
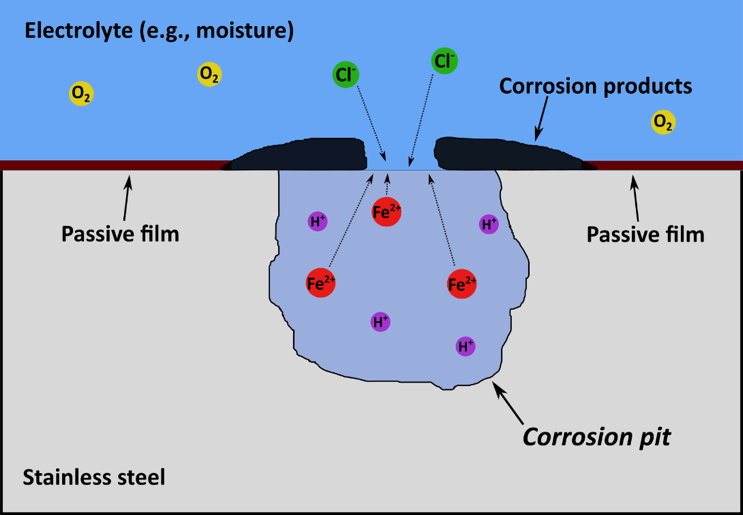 Figure 2 - Localized corrosion and Uniform Corrosion
Figure 2 - Localized corrosion and Uniform Corrosion
A schematic of the corrosion pit. Once the passive film breaks down a corrosion pit starts to grow. While the rest of the passive film remains intact and supports the cathodic reaction, the anodic reaction takes place inside the pit with a high dissolution rate.
Without passivity, most of the technologies that depend on the use of metals in any society could not exist. Passivity is a critical element in controlling the corrosion phenomena.
Once the passive breakdown happens, although the remainder of the passive film remains intact and continues to protect the metal against general corrosion, the localized corrosion proceeds at a considerably high rate because it is concentrated at a fixed area. Furthermore, compared to general corrosion, the presence of localized corrosion is more difficult to detect. The high corrosion rate and difficulty of detection, make localized corrosion dangerous as it can lead to catastrophic failure of the metal piece.
Crevice Corrosion Of Aluminium
Crevice corrosion of aluminium & it's alloys is relatively rare as compared to stainless steel it exhibits a relatively low susceptibility as the crevices tend to become filled with aluminium corrosion products.
Pitting Corrosion Of Aluminium
Pitting is one of the most destructive and treacherous forms of corrosion. During pitting corrosion of passive metals and alloys, local metal dissolution occurs leading to the formation of cavities within a passivated surface area, which causes an asset to fail because of perforation with only a small percent weight loss of the entire structure. Corrosion pits are sometimes isolated or in a cluster and can be small or large in diameter. In practice, pitting corrosion of nearly all passive metals and alloys is commonly observed in the presence of chlorides or other halides. Among engineering alloys, stainless steel suffers the most from pitting corrosion, although pitting has been frequently reported for Ni alloys, Al alloys, Cu alloys, and even galvanized steel. Also, pitting corrosion is one of the main corrosion forms that threaten the integrity of a pipeline. Furthermore, pitting corrosion can trigger other types of corrosion, stress corrosion cracking for example. Metallurgical factors, such as alloy chemical composition and microstructure, as well as environmental variables, like temperature and concentration of chlorides, directly affect the pitting corrosion resistance of an alloy. Because corrosion pits are often covered with corrosion products, it is generally difficult to detect them. Figure 2 below show all the details
 Figure 2 - Pitting Corrosion
Figure 2 - Pitting Corrosion
Galvanic Corrosion Involving Aluminium
Aluminium is low down in the galvanic series close to zinc and will corrode if in electrical contact with most other engineering metals such as steel, stainless steel & copper alloys. In these cells, aluminium forms the anode and the other metal the cathode. Relative surface area is a major issue so aluminium pop rivets will dissolve out very rapidly if used on copper components. The rate of galvanic corrosion depends both on the size of the driving voltage and the conductivity of the environment. Consequently, it is more of an issue in marine & industrial environments than rural ones. Figure 3 below shows an example
 Figure 3 - Galvanic Corrosion
Figure 3 - Galvanic Corrosion
Deposition Corrosion on Aluminium
Deposition corrosion occurs when a dissimilar metal gets deposited on the aluminium surface leading to serious localised corrosion.
Imagine water flowing through copper tubing. When the water flows through, it picks up copper ions. These copper ions are now in a solution. When this solution comes into contact with an aluminium surface or vessel, it deposits these copper ions onto it.
These ions now form a subtle galvanic cell which corrodes the aluminium through pitting if the ions are lower in the electrochemical or galvanic series. The larger the difference between aluminium and the deposited ion in the galvanic series, the worse the corrosion.
Even a concentration of 1 ppm copper ion solution is known to perform serious corrosion on the aluminium surface.
The metals that can cause deposition corrosion of aluminium are referred to as ‘heavy metals’. Some important heavy metals are copper, mercury, tin, nickel, and lead.
The corrosion caused by this method is more pronounced in acidic solutions as compared to alkaline solutions. This is because these ions have low solubility in alkaline solutions.For more details see figure 4 below
 Figure 4 - Deposition Corrosion
Figure 4 - Deposition Corrosion
End Grain Attack On aluminium
This phenomenon is similar to exfoliation corrosion. Figure 5 below shows an exfoliation corrosion. In both types of attack, the corrosion travels along a number of planes running parallel to the rolling direction and leaves areas or sheets of relatively unaffected material in between. The corrosion products tend to push the sheets of sound material apart. In materials science, intergranular corrosion, also known as intergranular attack, is a form of corrosion where the boundaries of crystallites of the material are more susceptible to corrosion than their insides.
Exfoliation corrosion is a specific type of selective attack that proceeds along multiple narrow paths e.g. grain boundaries which run parallel to the surface of a metal. Generation of corrosion products forces the layers apart and causes the metal to ‘swell’. Metal flakes may be pushed up and even peel from the metal surface. Exfoliation corrosion is most common in the heat-treatable Al-Mg-Cu and Al-Zn-Mg-Cu alloys. This form of corrosion is associated with a marked directionality of the grain structure. Applied stresses are not necessary for exfoliation to occur. However, in alloys susceptible to stress corrosion cracking, the corrosion product wedging action undoubtedly contributes to the propagation of the exfoliation attack. It is important to note that some alloys not susceptible to stress corrosion cracking (Al-Mg-Si) may suffer exfoliation corrosion. However, if the grain structure is equiaxed, exfoliation corrosion does not usually occur
 Figure 5 - Exfoliation Corrosion
Figure 5 - Exfoliation Corrosion
Filiform Corrosion
Filiform corrosion can occur in conditions slightly above room temperature and at a humidity level of 75%.
In places where filiform corrosion has occurred, a thread-like filament forms under the coating. The coating will bulge and have an appearance like that of a lawn riddled by mole tunnels. The filament will then continue to form until the coating is no longer continuous. Filiform corrosion can form with forming with a number of coating systems.
Filiform corrosion's unique appearance resembles fine filaments that look like worm-like threads and that seem to emanate from one or more defects in slightly random directions. Normally, it doesn't extensively damage the metal. However, it has a detrimental effect on the corroded metal's appearance.
For instance, filiform corrosion within magnesium or aluminium can form a white precipitate, resulting in a tail. On the other hand, filiform corrosion in iron tends to form a head with green fluid and a tail of red precipitate. Filiform corrosion often starts off as coating defects like scratches and weak points such as beards and holes.
A number of approaches have been known to reduce filiform corrosion. One of them is applying several coating layers. One might also use chromate containing a primer on aluminium or a conversion coating. Zinc with a primer on steel can also be used as a treatment. Figure 6 below shows an Filiform Corrosion
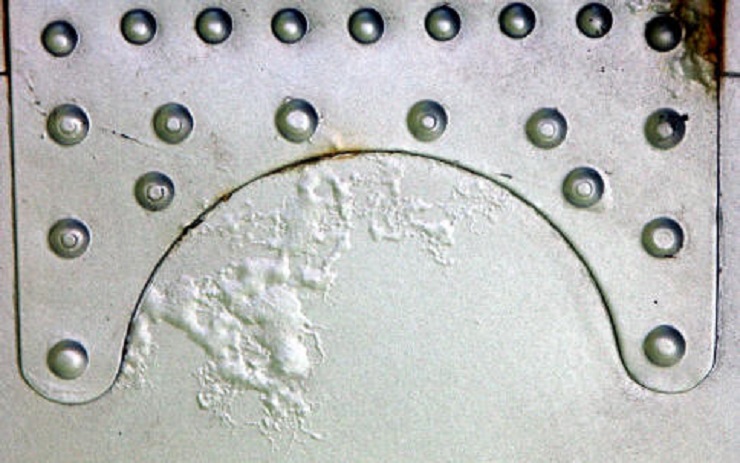 Figure 6 - Filiform corrosion
Figure 6 - Filiform corrosion
Surface Treatments & Surface Coatings For Aluminium (Solution)
The most common surface modification to aluminium surfaces is anodising. There are three common types of anodising treatment and they are commonly known as types I, II, & III. Type I anodising is carried out in Chromic acid & is relatively uncommon due to environmental reasons but still persists in the aerospace industry. Types II & III anodising are carried out in sulphuric acid. Type II is the most common type for decorative finishes & as a pre-treatment prior to the application of organic coatings. The car amplifier as shown in Figure 7 has been sealed using a dye in the hot water sealing solution, while, the keyboard in Figure 8 has been sealed without a dye in the sealing water.
|
|
|
working principle of aluminum welding machine
As a commonly used welding metal machine, the aluminium welding machine is small in size, light in weight and simple in operation, and is generally welcomed by the welding industry including the welders that operate it.
The aluminium welding machine adopts low-voltage and high-current electric energy to convert electric energy into thermal energy through the arc instantaneously. It uses high-purity argon gas as the shielding gas during welding to avoid the generation of pores and impurities during welding. At the same time, both AC argon arc welding and MIG gas-shielded welding. It has a certain cathode cleaning function, which can directly remove the oxide film on aluminium and aluminium alloy. Because the aluminium welding machine is small in size, simple in operation, convenient in use, high in welding efficiency, good in weld formation, large in penetration depth, capable of welding aluminium and aluminium alloy plates to achieve excellent bonding effect, and the welding strength is the same as that of the base metal, and the sealing is the same. It is very good and is widely used in various fields of industry and life.
The aluminium welding machine will produce arc light during use. The arc contains infrared rays and ultraviolet rays. At the same time, it also produces harmful substances such as metal vapour and smoke. The tungsten rod in tungsten argon arc welding contains a small amount of radioactive elements, so it must be protected. Measures, in addition to the use of argon as a shielding gas, should not be operated in a windy welding field. With the development of industrial technology, digital welding machines such as a single-chip microcomputer, DSP, FPGA, etc. have matured. The welding of special planes or arc welding robots through point-to-point and bus structures has also been applied to production one after another, which can replace the high difficulty that cannot be completed by humans. High-strength automatic welding operation, while digital aluminium welding machine supports network remote monitoring and fault diagnosis; the automation of aluminium welding machines will be widely promoted and applied in recent years.
Electrical Conductivity and Resistivity for Aluminum
The electrical conductivity of 99.99% pure aluminium at 200C is 63.8% of the International Annealed Copper Standard (IACS). Because of its low specific gravity, the mass electrical conductivity of pure aluminium is more than twice that of annealed copper and greater than that of any other metal. The resistivity at 200C is 2.69 micro ohm cm.
The electrical conductivity which is the reciprocal of resistivity, is one of the more sensitive properties of aluminium being affected by both, changes in composition and thermal treatment. The addition of other metals in aluminium alloys lowers the electrical conductivity of the aluminium therefore this must be offset against any additional benefits which may be gained, such as an increase in strength.
Heat treatment also affects conductivity since elements in solid solutions produce greater resistance than undissolved constituents. The very good electrical properties of aluminium have made it an obvious choice for applications in the electrical industry, particularly in power distribution where it is used almost exclusively for overhead transmission lines and busbars. The first major aluminium transmission line was completed in 1898 in the USA: a 46-mile, three-phase installation for the Standard Electric Company of California, from Blue Lakes to Stockton.
Its use later became much more general when it was found possible to reinforce the cable (usually alloy 1350) with galvanised steel wire which increased the spans without too much sag. Although this product is still used, high-strength (6061 type) aluminium multi-strand cables are now preferred for some installations because higher line tensions can be achieved which can be applied to increase the distance between the pylons or alternatively reduce their height.
Electrical conductivity is one of the most sensitive properties of aluminium, being particularly responsive to changes in composition and temper. All known metallic additions, reduce aluminium's electrical conductivity. Metals in solid solution depress the conductivity to a greater extent than when out of solution, which means that the temper of an alloy has an influence of the conductivity. Pure aluminium (Al99,99) has an electrical conductivity of 37,67 m/Ohm mm2 (= 64,94% IACS) at 20°C, which corresponds to an electrical resistivity of 26,5 nm. Commercial wrought aluminium alloys show thermal conductivity values in the range of 15ñ36 m/Ohm mm2 at 20°C, depending on composition. Cast aluminium alloys vary for the same reason from 10ñ32 m/Ohm mm2.
What Makes
Why Choosing Us
30 Years of Experience
Award Winning
Warranty
800+ Sucessful Project
Honest & Dependable
Professional Workers
Request for Quote
Hot ! Deal With Factory Plus and
Get More Valuable Services
Explain to you how all this mistaken ideas of denouncing pleasures
and praising pain was born and I will give you a complete account of
the system.
For Business: For Business inquiry pls fill our feedback form and
Troll Free Number – (+91) 00-700-6202.
About
We offer the best and economical solutions, backed by 27+ years of experience and international standards knowledge, echnological changes, and industrial systems.

























