Composition and characteristics of LPG (liquefied petroleum gas)
Butane, Propane, LPG: let's clarify. How many of you did it happen that at shallow temperatures, perhaps after a day of skiing, the heating suddenly stopped working? So here is a practical guide to understand what happened and then make sure it doesn't happen again We can say that safely; the gas does not freeze. At least, it does not freeze at even the most extreme temperatures found in nature on our planet. The list below shows the gas solidification temperatures:
The table above with the melting (or freezing, if you prefer) temperatures of some gases are therefore all around -150 ° C or even more extreme. They are values established at a pressure of 1 bar but, at the pressure of 5 or 6 bar of our cylinders, it changes very little. For those who want to remove the doubt and do some calculations, this is the formula that relates the freezing point of propane with pressure and temperature: P=-718+2.38565xT^1.283 where P is the pressure in bar and T is the temperature in Kelvin. I just remember that 0 ° K = -273.15 ° C and that propane freezing occurs at -187.69 ° C = 85.46 ° K The table below shows the automatic temperature conversion Propane and Butane Having clarified that the gas cannot freeze, it remains to understand why butane leaves us in the cold when the outside temperature approaches 0 ° C, while propane continues to do its duty even with shallow temperatures (for us humans). The "secret" is in the different boiling temperatures of the two gases, i.e. the temperature below which the gas remains liquid and cannot evaporate to transform itself into a ready-to-burn gas. The boiling temperature for butane is -0.4 ° C while for propane it is -43 ° C. This means that if we have a cylinder full of butane and the temperature drops to 0 ° C or less, the air will not give enough heat to the cylinder to evaporate the gas. The gas remains liquid, and the flame goes out. If we have a propane cylinder and the temperature dropped even to -20 or -30 ° C, the air would still be able to transfer heat to the cylinder, thanks to the temperature difference, guaranteeing the evaporation of the liquid gas and therefore the functioning of combustion appliances. However, if the outside temperature dropped up to -40 ° C, even the propane would have difficulty evaporating as it no longer received heat from the outside and the combustion also in this case would cease. If we have a "double" mixture in the cylinder, at temperatures above -40 ° C, it will be possible to use only the propane part, then when this is exhausted, and only a butane residue remains, the flame will stop. There is an ingenious way to solve this problem, heating the gas as shown in Figure 2 below
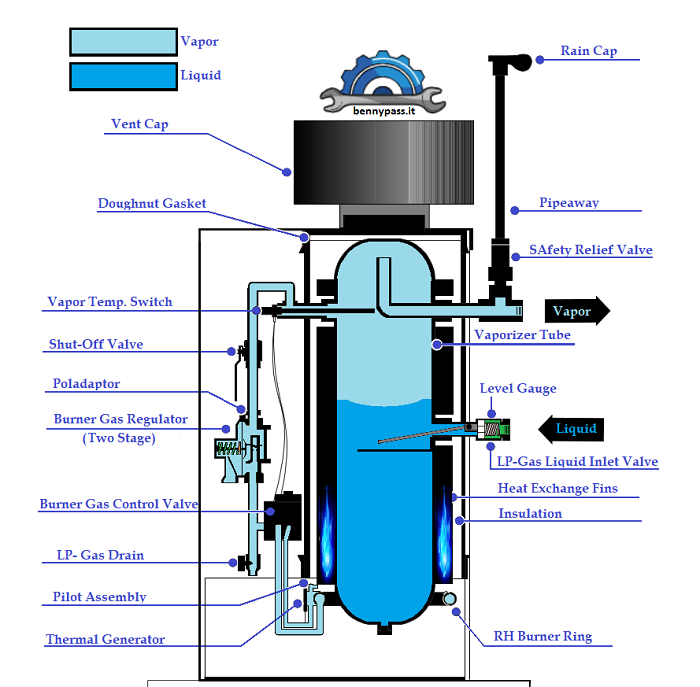
For more details, read the Propane Vaporizers
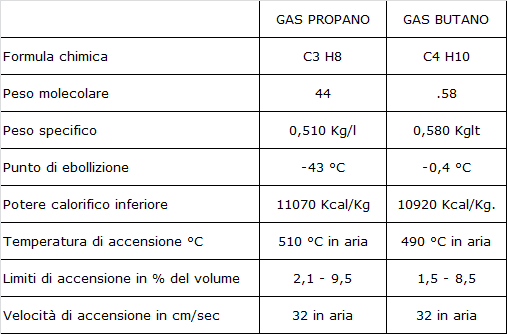
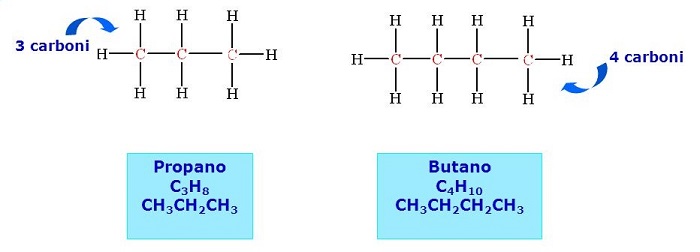
LPG The name of LPG (liquefied petroleum gas) indicates those blends of commercial butane and commercial propane obtained from the distillation of crude oil, from the fractionation of natural gas (which they are often contained) and from the processing of petrochemical plants. At ambient temperature and atmospheric pressure, they occur in the form of gas. Commercial Propane can be mixed with propylene, butylene, butane and ethylene. The Commercial Blend is a comprehensive set of hydrocarbons, obtained from the combination for the most part (about M 70%) of butane and isobutane and with an amount of propane equal, on average, to 30%. Suitably odorized and denatured, this mixture is sold as a domestic fuel in cylinders. Figure 3 -4 below shows the physical and chemical properties
Main characteristics of Butane and Propane One of the main characteristics that distinguish butane and propane, which determines their use, is the vapor pressure which corresponds to the pressure of the gaseous phase in equilibrium with the liquid phase in a closed container. The vapor pressure determines considerable variations in the pressure of the mixture as the percentage of butane and propane varies. This pressure also increases with increasing temperature and therefore leads to strong variations in the volume of LPG in the liquid state. Therefore, if a container is full of L.P.G. in the liquid phase and the temperature continues to rise, there is a rapid increase in pressure, which can also cause the container to burst. It is essential never to fill with L.P.G liquid the container. Fortunately, existing the relief valves that open in case of overpressure (where they are present) Another essential characteristic differentiating the two types of gas (butane and propane) is the boiling point, i.e. the temperature at which the liquid state changes to the gaseous state. While propane at a temperature of -43 degrees centigrade no longer gases and remains liquid, for butane, this occurs at a temperature of -0.4 degrees centigrade. This determines the need to use, in freezing climates, mixtures with pretty high percentages of propane, to favour gasification. A peculiar characteristic of the L.P.G. is dissolving fats, oils, paints. It also causes the deformation of natural rubber. Therefore the flexible pipes that make up the vehicle system are made of synthetic material of adequate quality. |
About
We offer the best and economical solutions, backed by 27+ years of experience and international standards knowledge, echnological changes, and industrial systems.